Leave a comment at the end of this page or email contact@krishagni.com
v8.0 Release Notes
Introduction
We are happy to announce that v8.0 is ready for download! This release includes some important bug fixes and enhancements requested by customers.
New features include:
- Quick Access Widgets on Home page
- New Forms user interface
- Record User Training data (Univ of California Davis)
- Query enhancement to limit total quantity of specimens in results (University of Pittsburgh)
- Survey enhancements (Columbia University Medical Center)
- Supplies enhancements to support bulk import (Quantum Leap / Univ of California SF)
Download
Version | Link | Questions |
|---|---|---|
| Enterprise | Contact support team | |
| Community | Source Code GitHub link | forums.openspecimen.org |
Software/Hardware requirements
Review this page before attempting to upgrade for any changes: Software/Hardware Requirements.
Quick Access Widgets on Home page
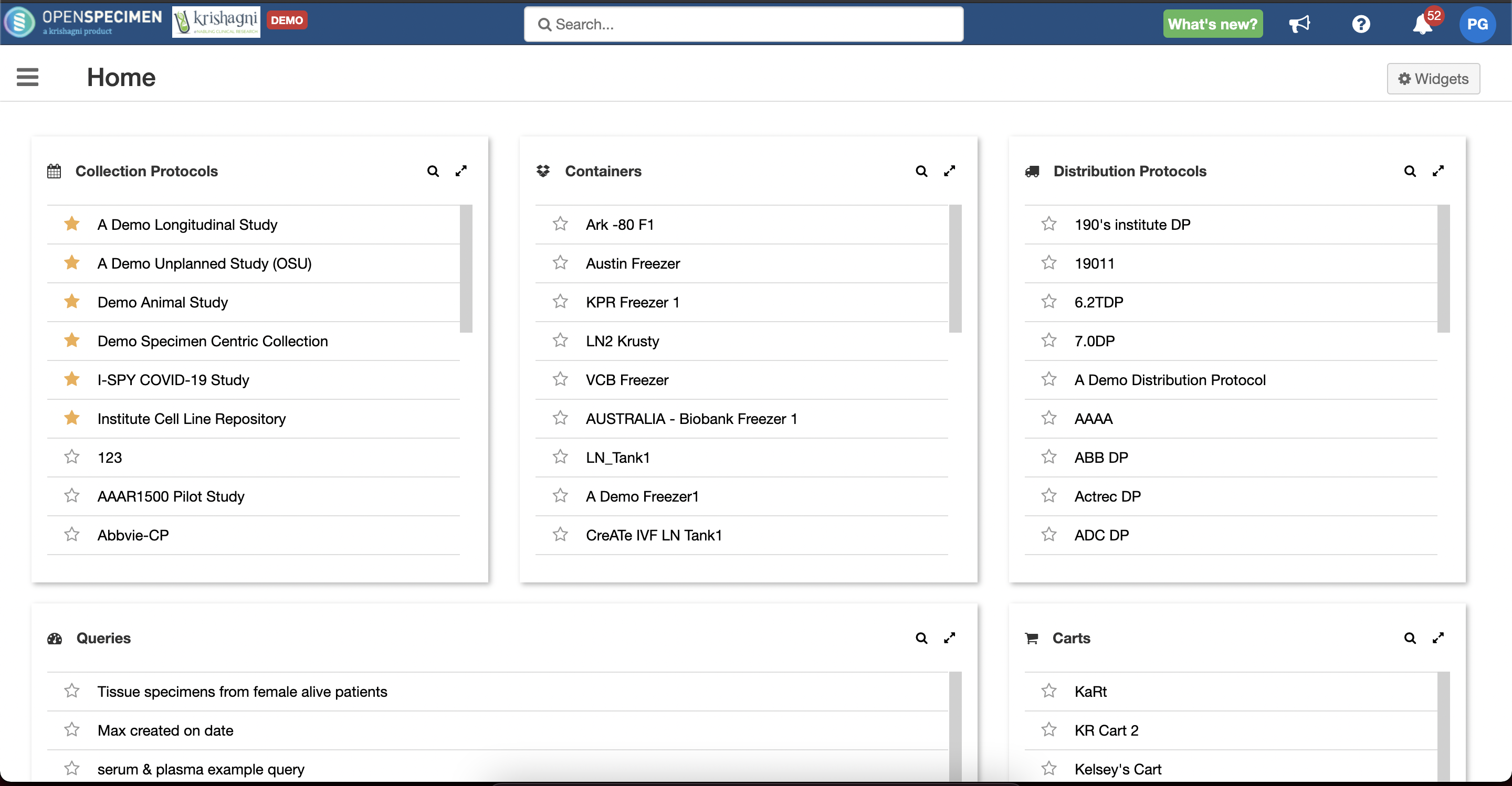
The Home page can now be customized with "quick access" widgets. Within a single click, you can navigate to the object you are looking for.
You can select what widgets to display for your account like list of protocols, queries, orders, forms etc.
Within these lists, you can set favorites to be displayed at the top.
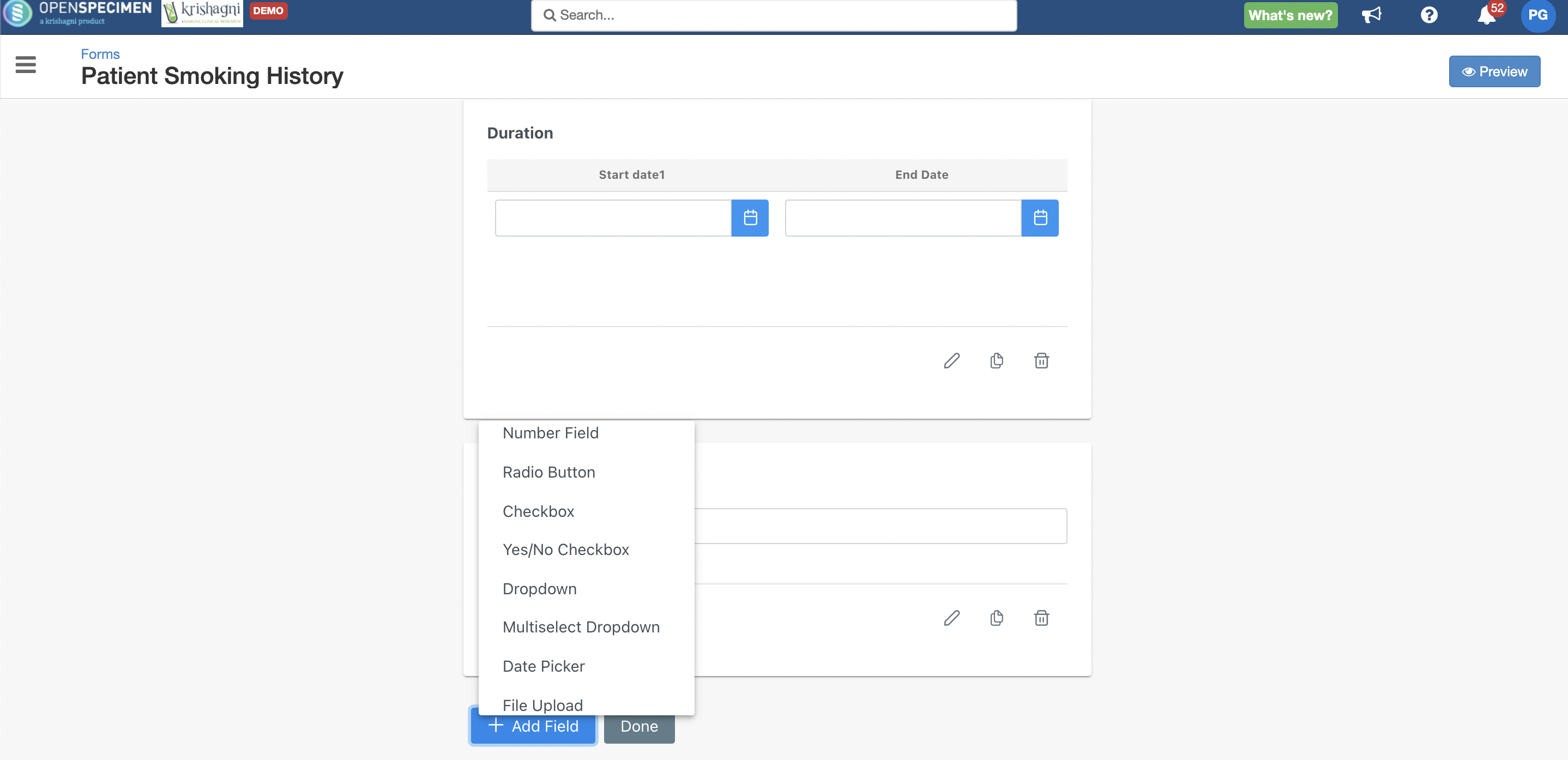
New Custom Forms UI
Forms creation interface is redesigned to make it more user-friendly and to improve performance.
For more details, refer to Custom Forms
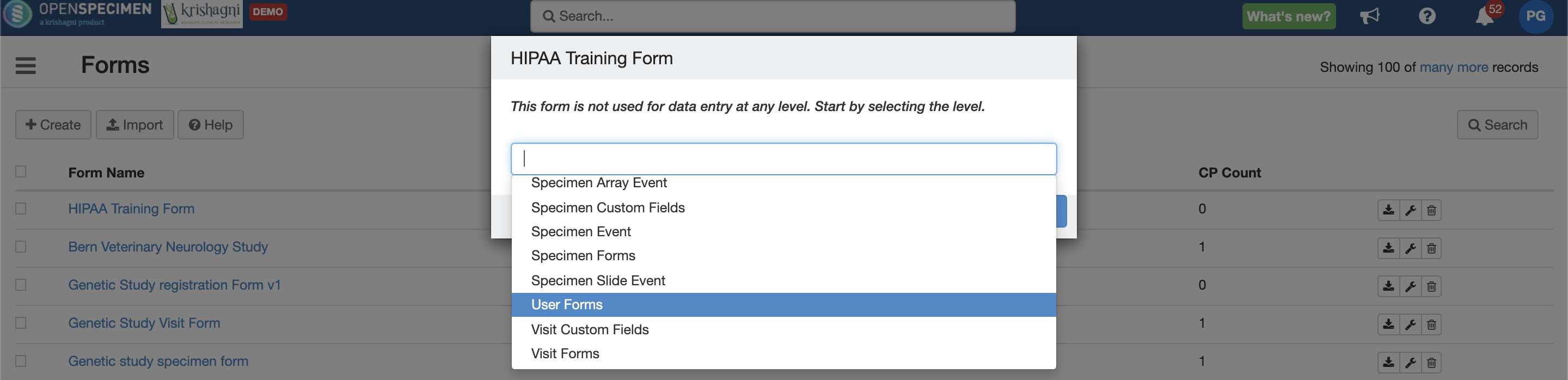
User-level Forms
Admins can now create forms and attach at user level. This is useful to track any additional information related to users like training forms.
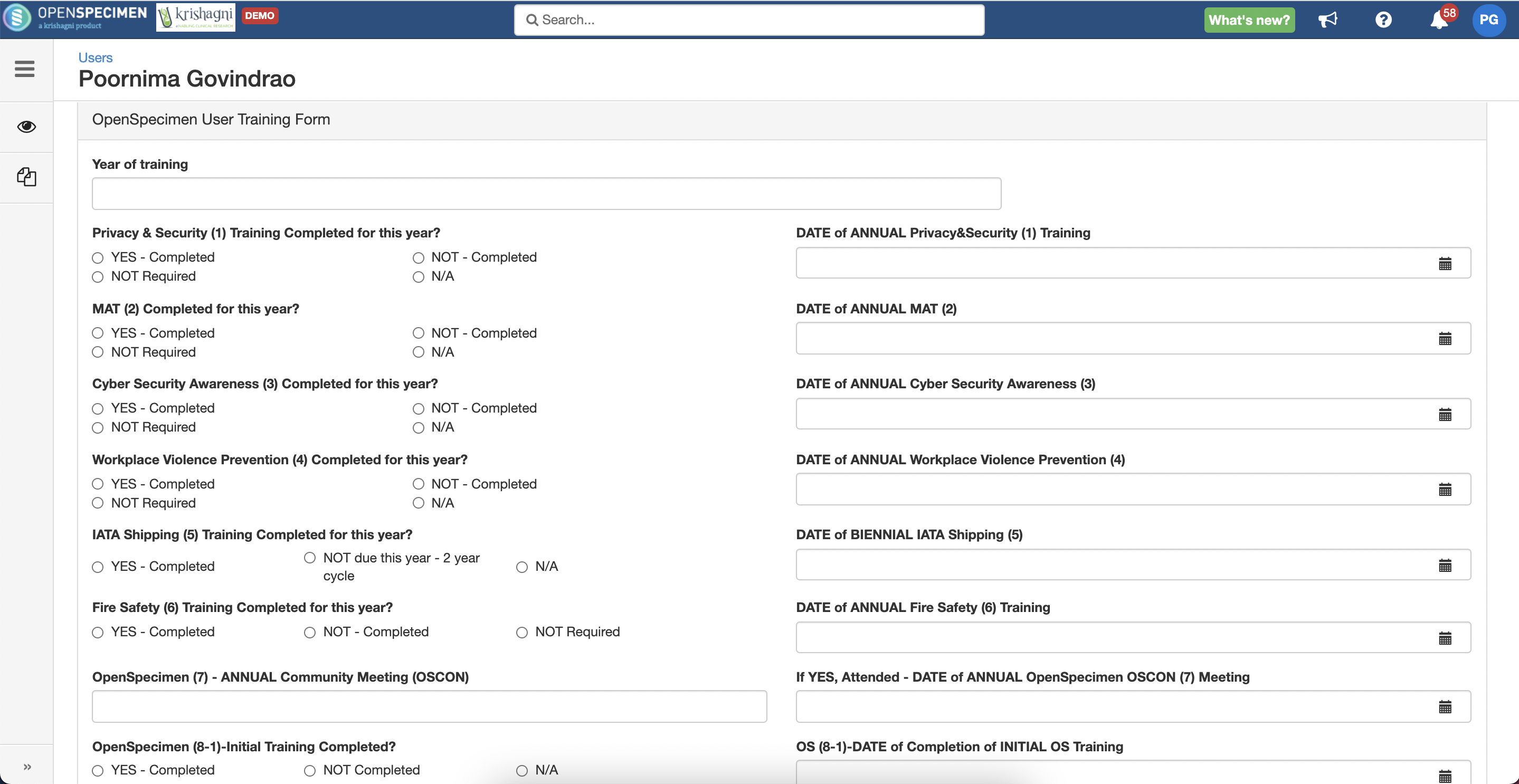
User Training Records
Using the feature of 'User' level custom forms, now it is possible to track user's training data. For e.g. HIPAA training. As part of v8.0, a default user training form is available within OpenSpecimen. Admins can reuse this or edit as per local requirements.
Supplies
Inputs from Quantum Leap Healthcare Collaborative / Univ of California SF
Below are some enhancements implemented in Supplies module:
Ability to select one or more supply items and create a new supply with the selected items or move it under existing supply. This can be used to move supplies from one site to another.
Ability to bulk import and export - supply types, supplies and supply items.
Ability to automatically mark expired supply items as "Expired" after the buffer/grace time is completed.
Survey Enhancements
Inputs from Columbia University Irving Medical Center
Survey reports can now be generated using queries module. This allows users to generate reports based on any Survey fields as well as create summary reports and schedule it to run automatically.
For more details, refer to 'Surveys'
OpenClinica Enhancements
Inputs from FIND Diagnostics
You can now ignore participants based on site ID of the subject in OpenClinica when syncing data from OpenClinica to OpenSpecimen. This is useful when the OpenClinica project is used for multiple sites but the OpenSpecimen is used by a subset of the sites.
E.g. A single OpenClinica server is used by both two clients, but they have their own OpenSpecimen instances.
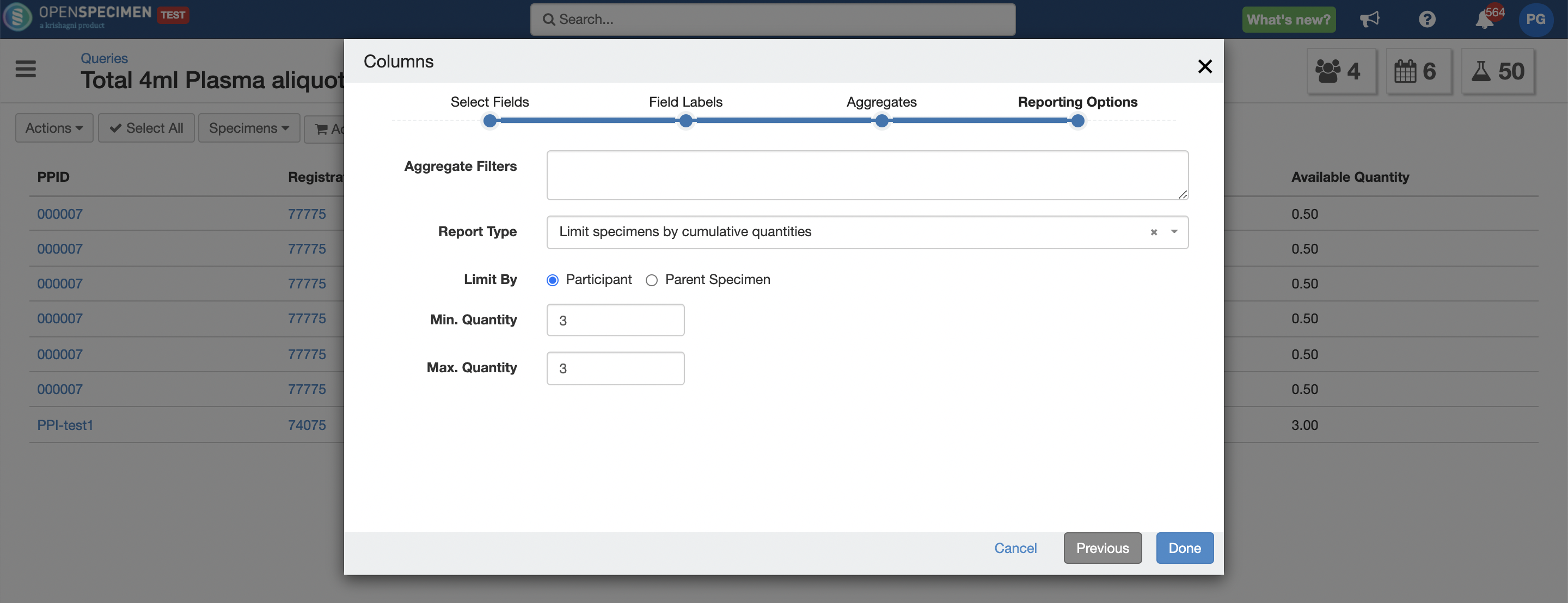
Query Enhancements
Below are some enhancements implemented in Queries module:
- Ability to search for more than 1000 values when adding criteria on text fields like MRN, label, barcode etc. using 'is one of' and 'is not one of' operator.
- Ability to limit specimens based on total quantity of aliquots by parent or patient. For e.g. Find all available aliquots with total quantity of 20 ml plasma per patient.
Bugs & Enhancements
Below are some other fixes implemented in v8.0:
| Description | Reported by |
|---|---|
- Able to specify the custom size of lists cart, specimens, participants, containers, protocols etc.
- Ability to return specimens with zero quantity
- Ability to edit system events like transfer event, disposal event, reserve events etc via bulk import.
- If there are deviations like missed specimens or decreased number of aliquots, users are prompted to re-run auto allocation.
- Ability to update barcodes to specimens based on specimen locations: This is useful for users who use flatbed or box scanners.
All Improvements and Bugs
Needs Jira login to view the below table.
Related content
Leave a comment at the end of this page or email contact@krishagni.com