Leave a comment at the end of this page or email contact@krishagni.com
Bulk Registration and Kit Management
Introduced in v4.0
Introduction
In high throughput biobanks where samples are generally collected across multiple sites, the biobankers wish to be ready with specimen collection kit before collection. To facilitate this, OpenSpecimen supports below features:
- Bulk registration of participants
- Specimen kit management
Pre-requisite
Set autogeneration for participant protocol identifier(PPID) and the specimen labels. For more details on label configuration and tokens, please refer to page
Enabling the bulk registration
At the protocol level, administrators can enable bulk registration. This will allow tissue bankers to register multiple participants at a time. The PPID format is mandatory to enable this feature since PPID is mandatory field during registration.
In order to enable this-
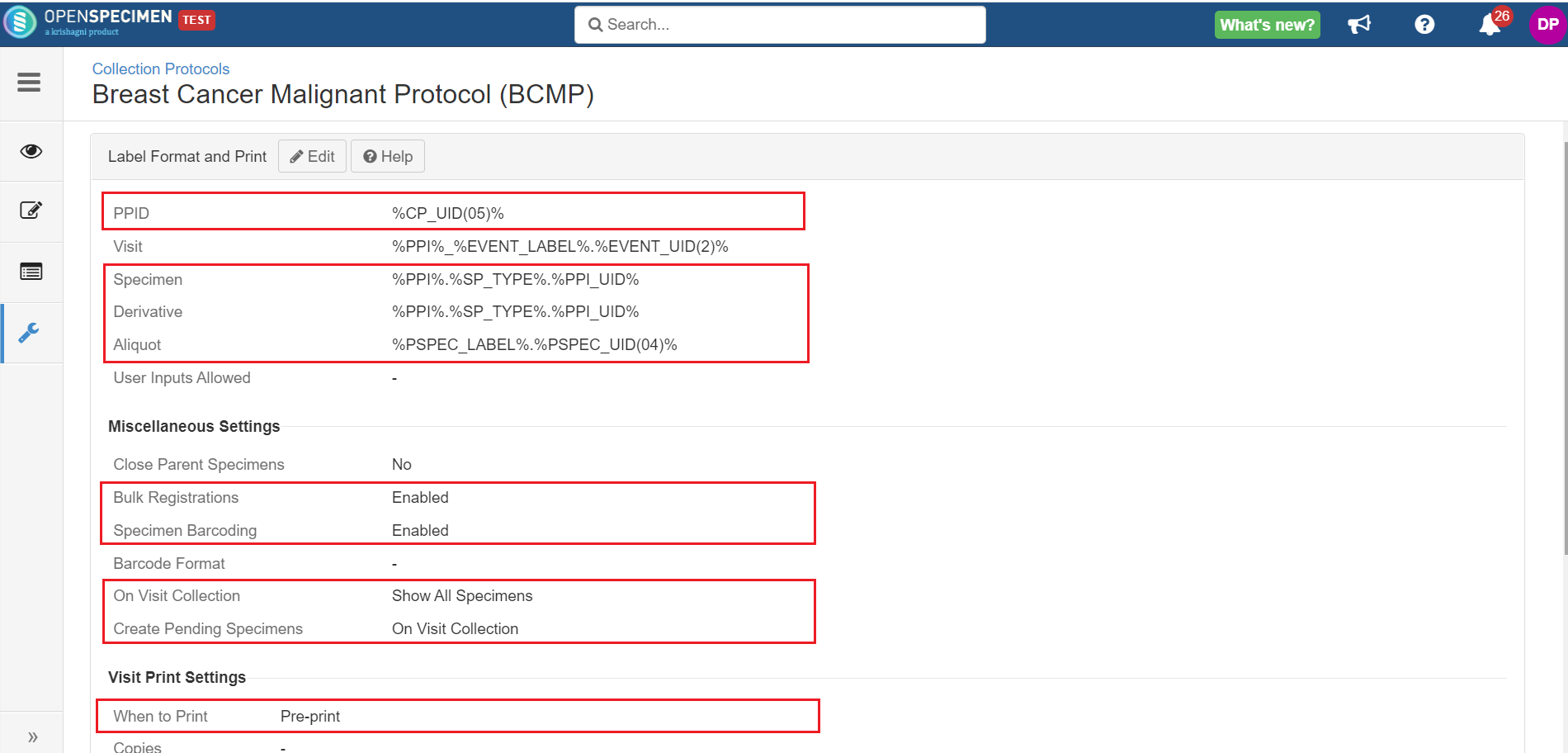
In order to enable 'Kit Management,' enable 'Pre-print Labels' and select appropriate settings for 'When to print.' This will create a kit with specimens selected for pre-print.
For example, as per setting in the above screenshot, all aliquots will be created as part of the kit on the creation of visit.
Bulk registration and Kit creation
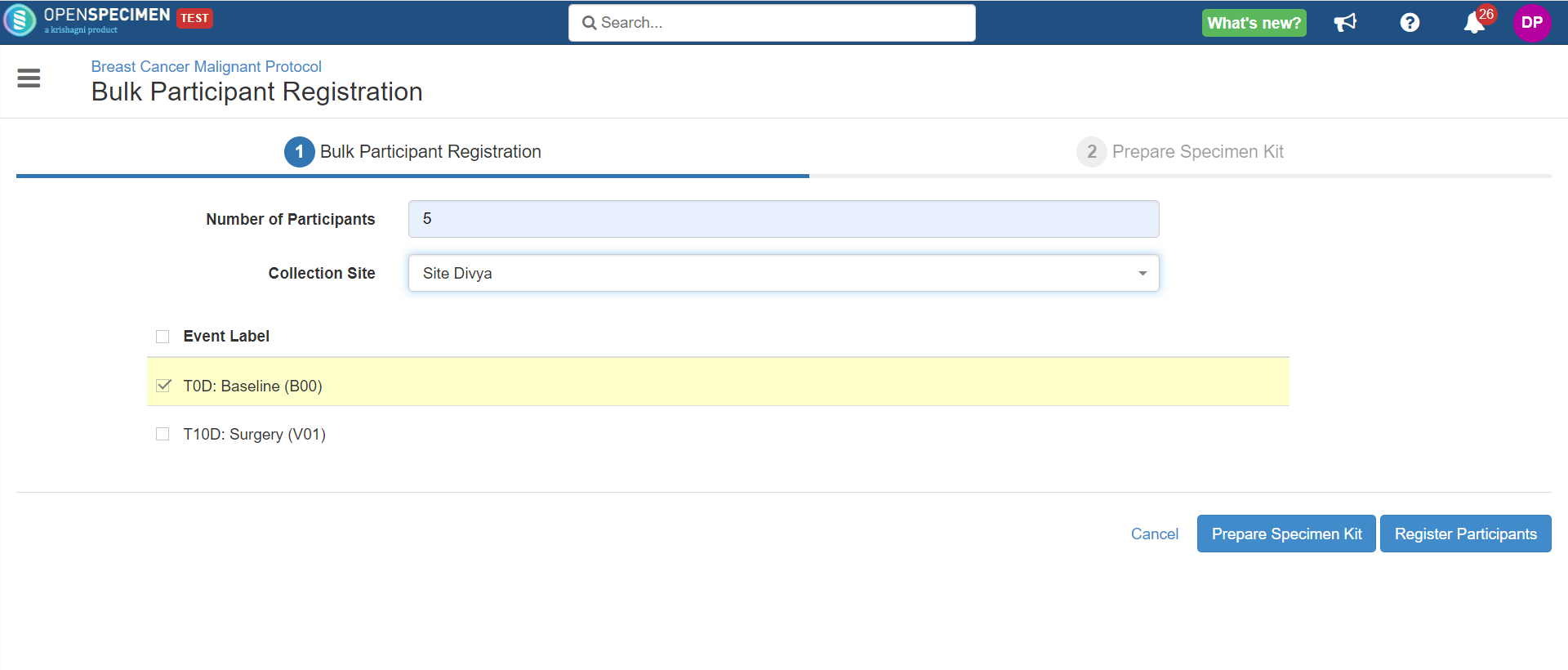
To register multiple participants, go to 'Participants' view of the specific protocol where 'Bulk Registration' is enabled. Click on 'Add Participant' → 'Multiple'
Enter number of participants to be registered along with collection site. Select the visit under which the specimens are being collected.
Note:
The "Collection Site" dropdown will only display the sites that are associated with the particular Collection Protocol.
Click on 'Register Participants' to create participants without specimen kit.
Click on 'Prepare Specimen Kit' to register participants, pre-print labels as per protocol setting, and create kit.
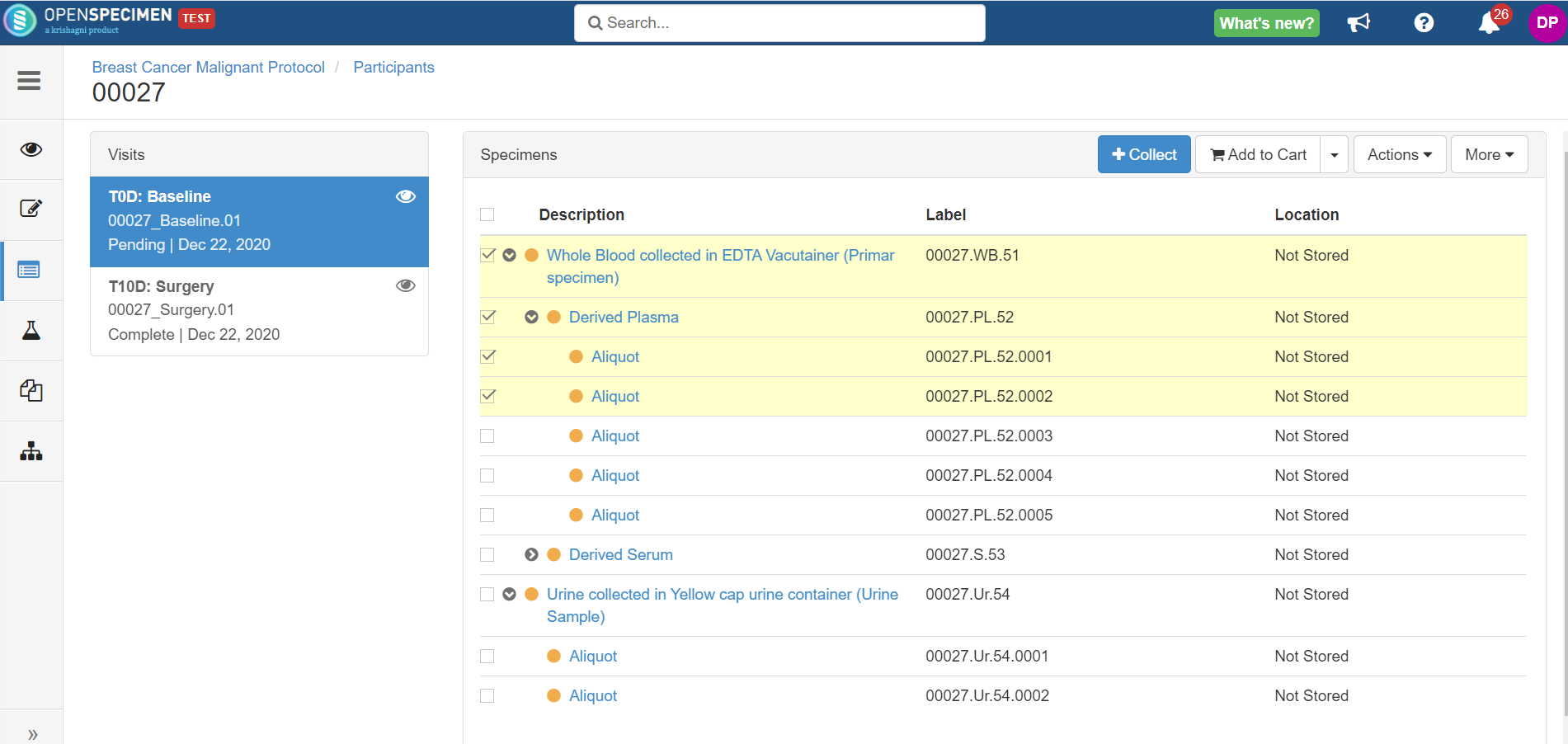
The participants are registered with appropriate auto PPIDs, labels, etc. You can collect the samples as per actual collections.
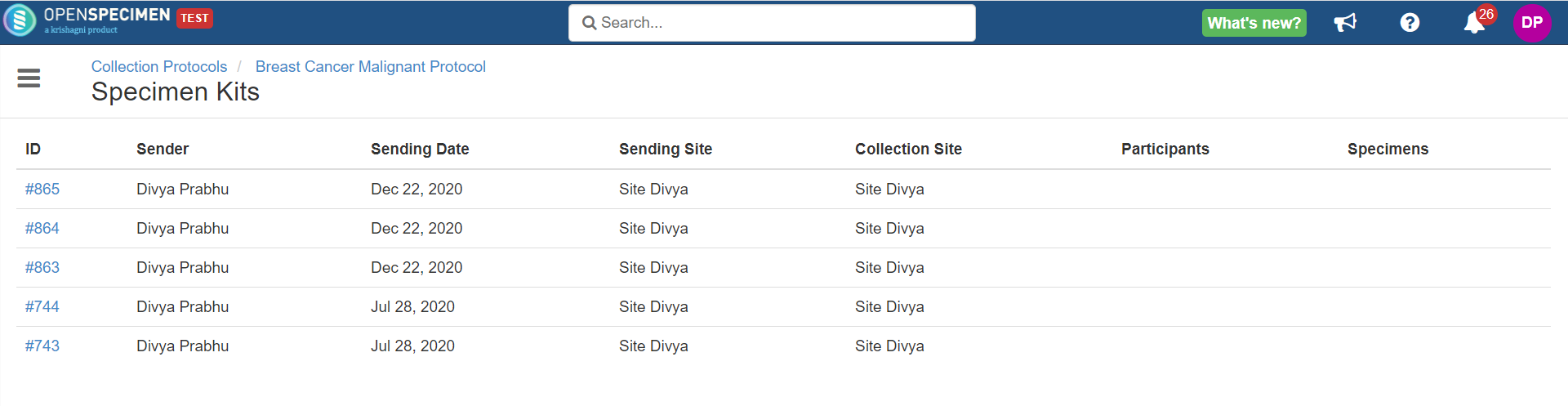
Specimen kits created can be accessed from 'Participants' view → 'More' → 'Specimen Kits.'
CSV report can be exported for every specimen kit; click on 'Download Report' button from specimen kits overview page.
Leave a comment at the end of this page or email contact@krishagni.com