We are happy to announce that v7.0 is ready for download! As always, it has new features, improvements and bug fixes selected based on feedback from many of our current adopters.
New features highlights:
Version | Link | Questions |
|---|---|---|
| Enterprise | Contact support team | support@krishagni.com |
| Community | Github repository for v7.0 | forums.openspecimen.org |
Software/Hardware requirements
Review this page before attempting to upgrade for any changes: Software/Hardware Requirements.
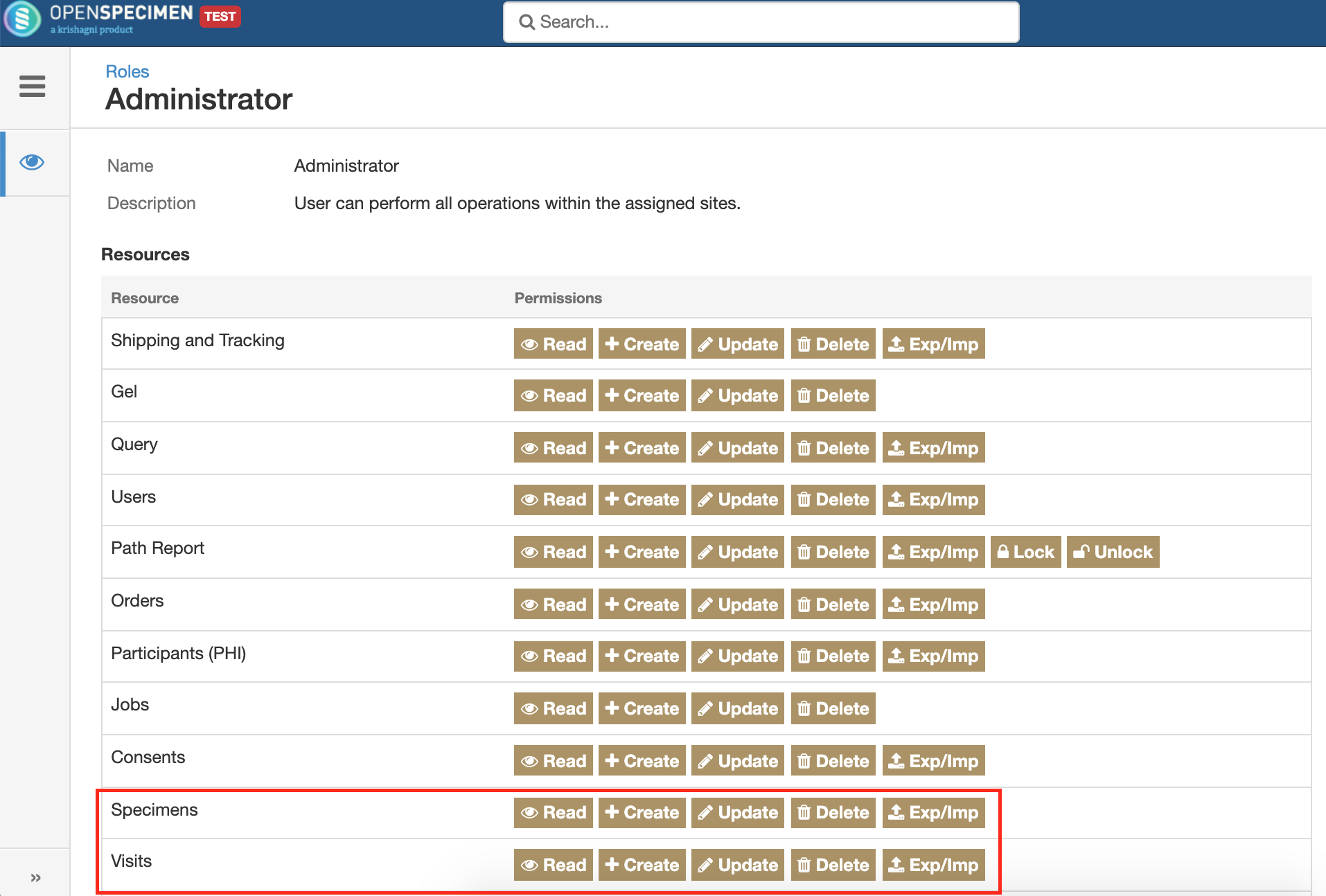
The visits and specimens resources under roles have been separated out. This will allow users to be given access to visits but not any specimens. This is useful if clinicians are only handling participant and visits.
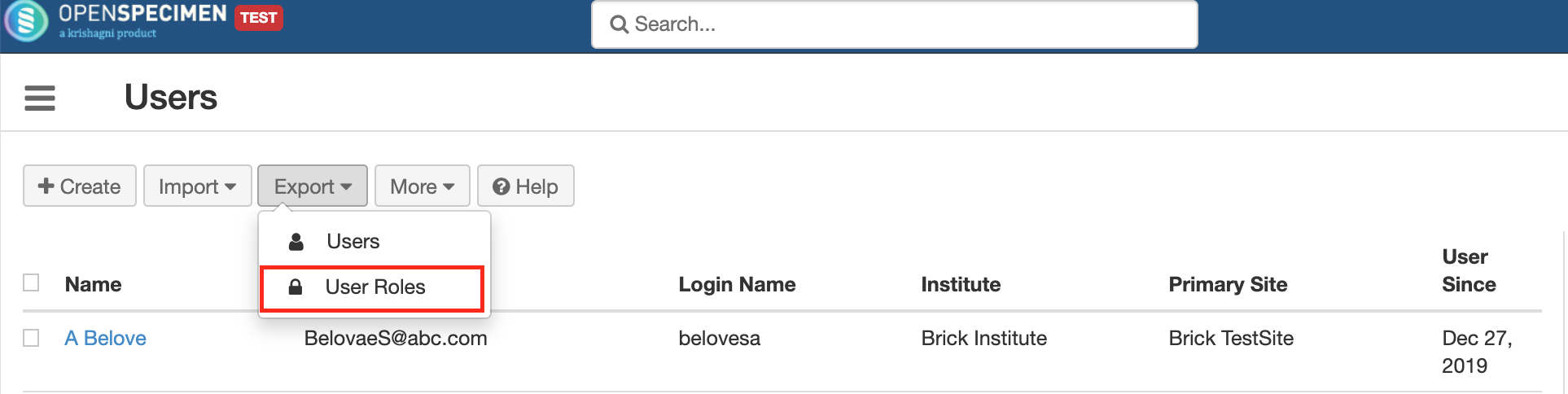
This helps in exporting user accounts and their roles to be imported to another server. For e.g. for moving user accounts from test to production server.
Using bulk operations, admins can now delete users, sites, institute and distribution protocols by setting activity status as 'Disabled'. Previously in this bulk templates, activity status column was missing.
Multiple specimens can now be edited at once via user interface. This is now possible for any field of specimen including custom fields using the 'Actions' menu present in multiple places like cart, container specimens view, specimens list within collection protocol, visit overview page etc.
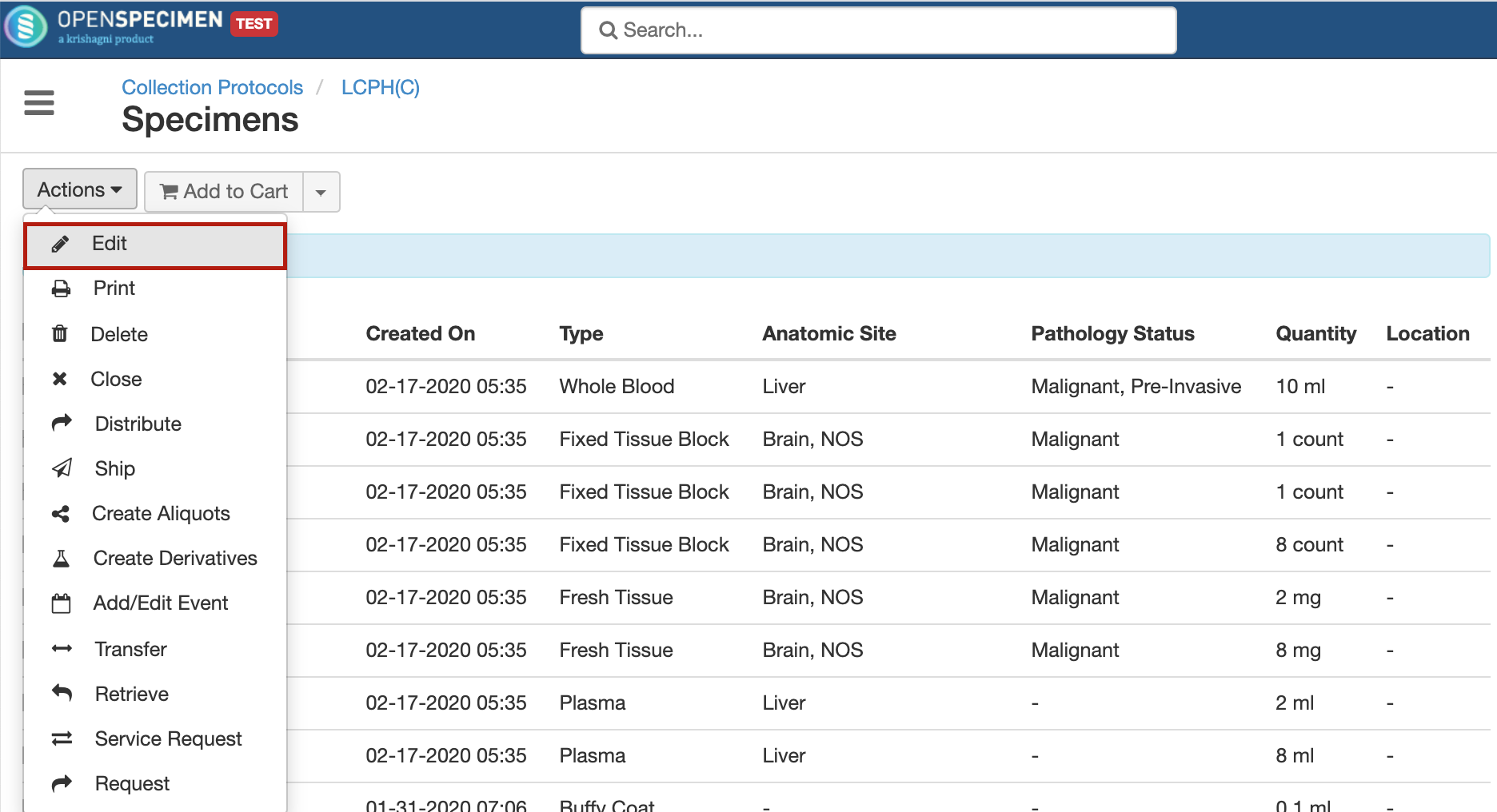
For more details, refer to 'Bulk Operations On Specimens'.
Multiple participants can now be updated or deleted via user interface from participants list within a collection protocol.
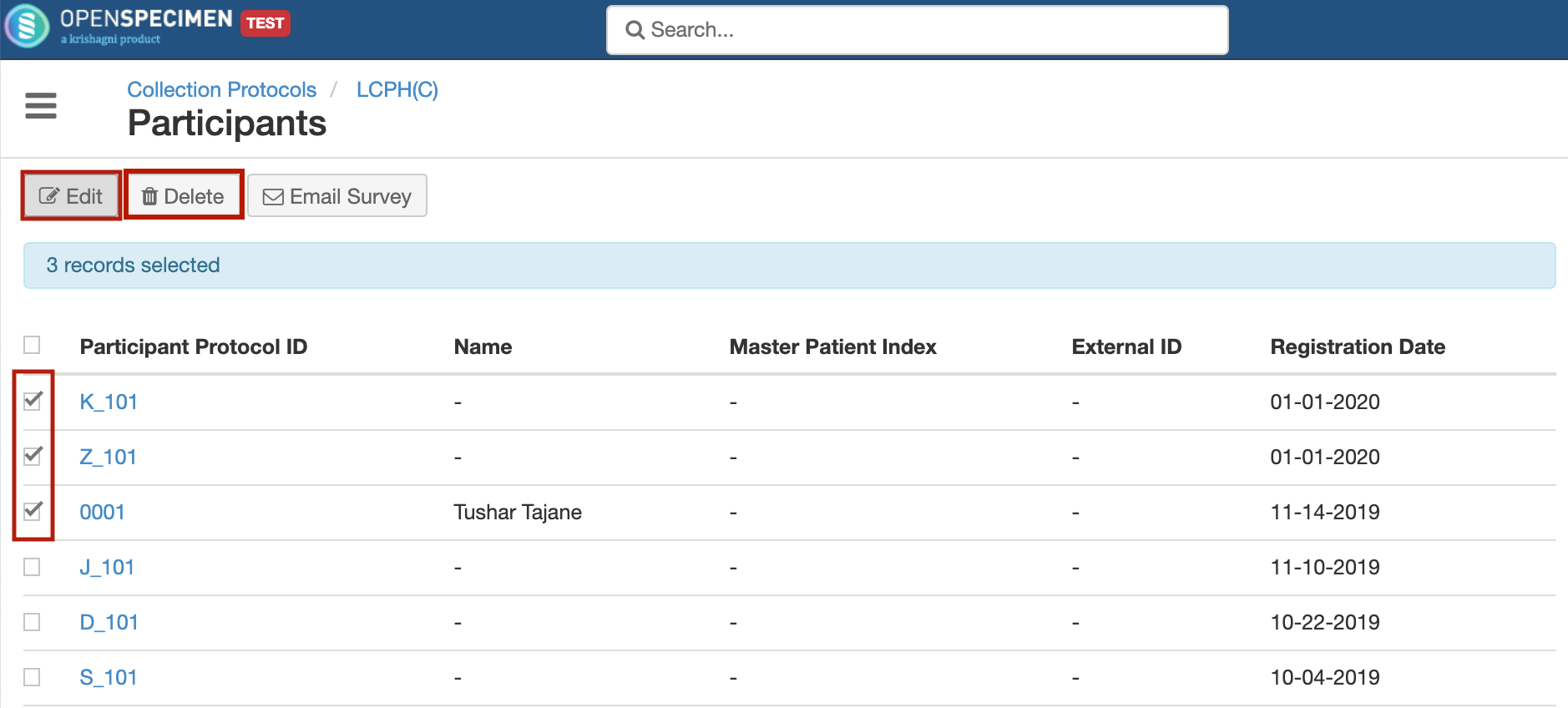
For more details, refer to 'Bulk update participants'.
Customs forms can be hidden/shown on the basis of rules defined in the JSON. This is useful to display specific forms for specific use cases.
For example:
Display "Tissue Review Event" only for tissue specimens
Refer to 'Hide/Show Custom Forms based on Rules (v7.0)' for more details.
When creating unplanned specimens, there is now option to create aliquots under the derivatives in a flow.
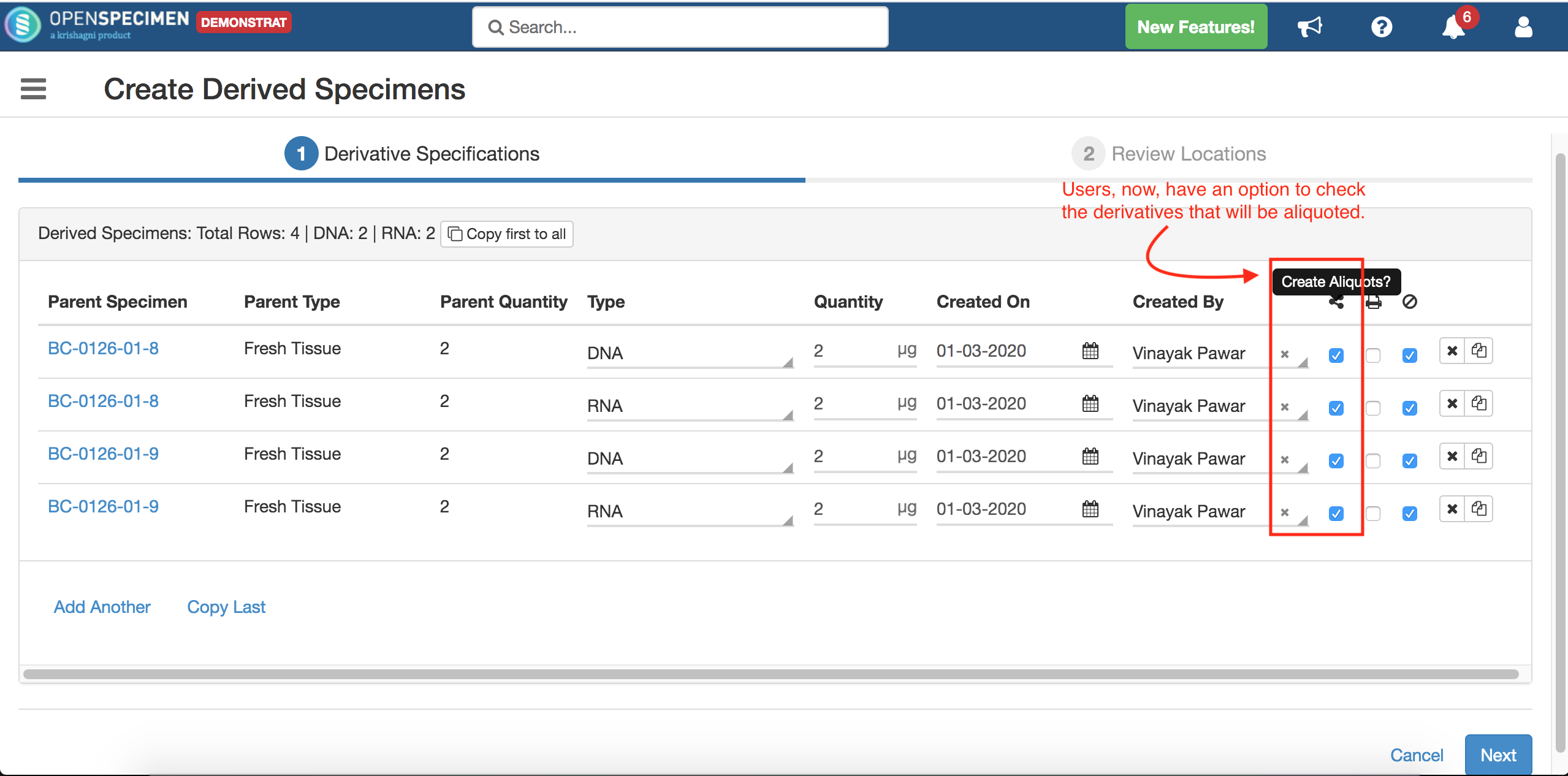
For more details, refer to 'Create aliquots from derivative page'.
OpenSpecimen's eConsents module helps clinical research centers to collect both types of consents - study-specific and broad-based consents. Below are the features of this module:
Create consent forms in IRB approved format
eSignature support
Consent form versioning
Multiple languages support
Supported on tablets
Reporting

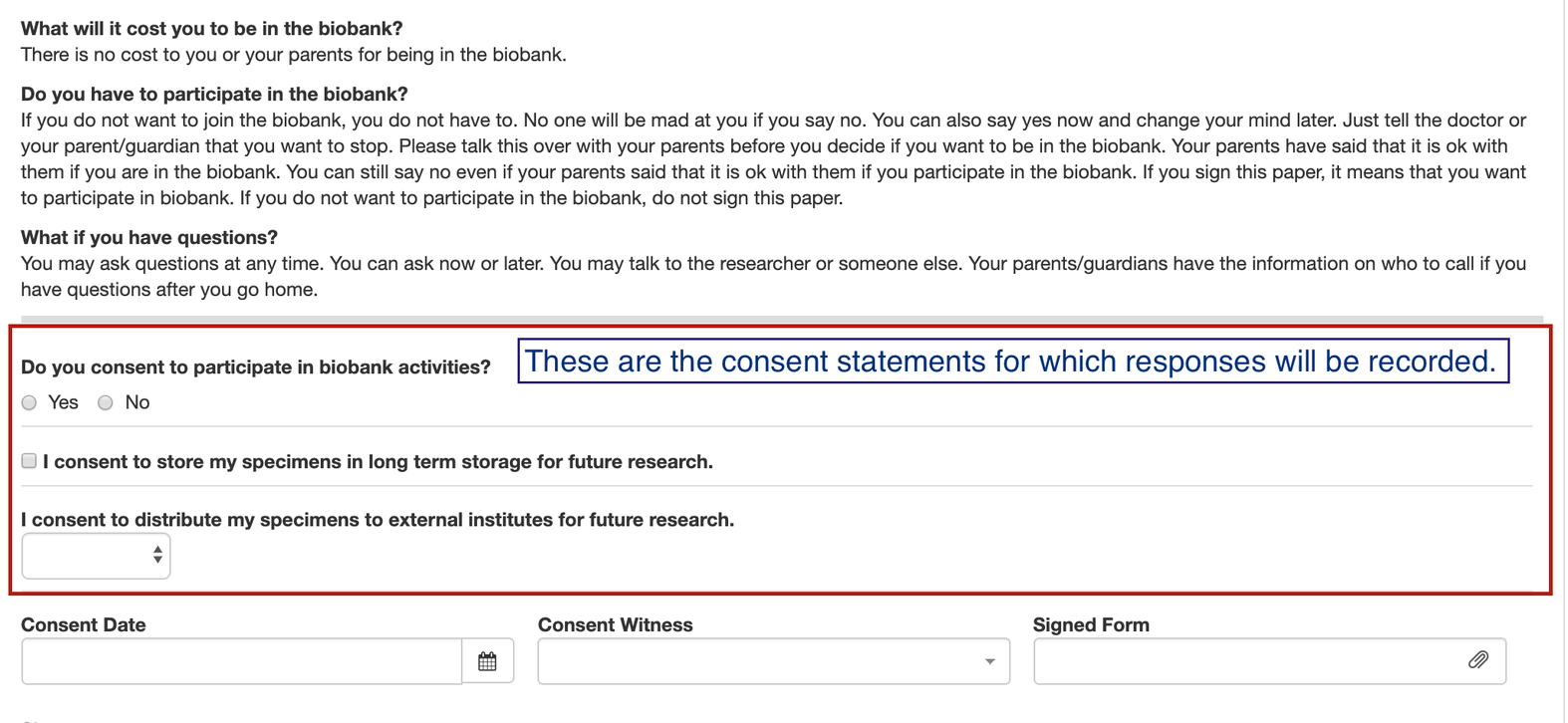
For more details, refer to 'eConsents'.
Forms (like consent forms, surveys, questionnaires, medical history, clinical history, etc.) can be collected directly from participants via email. Data gets recorded in OpenSpecimen once the patient clicks on the link and enters the data. This allows the patients to enter data at their convenience from anywhere.
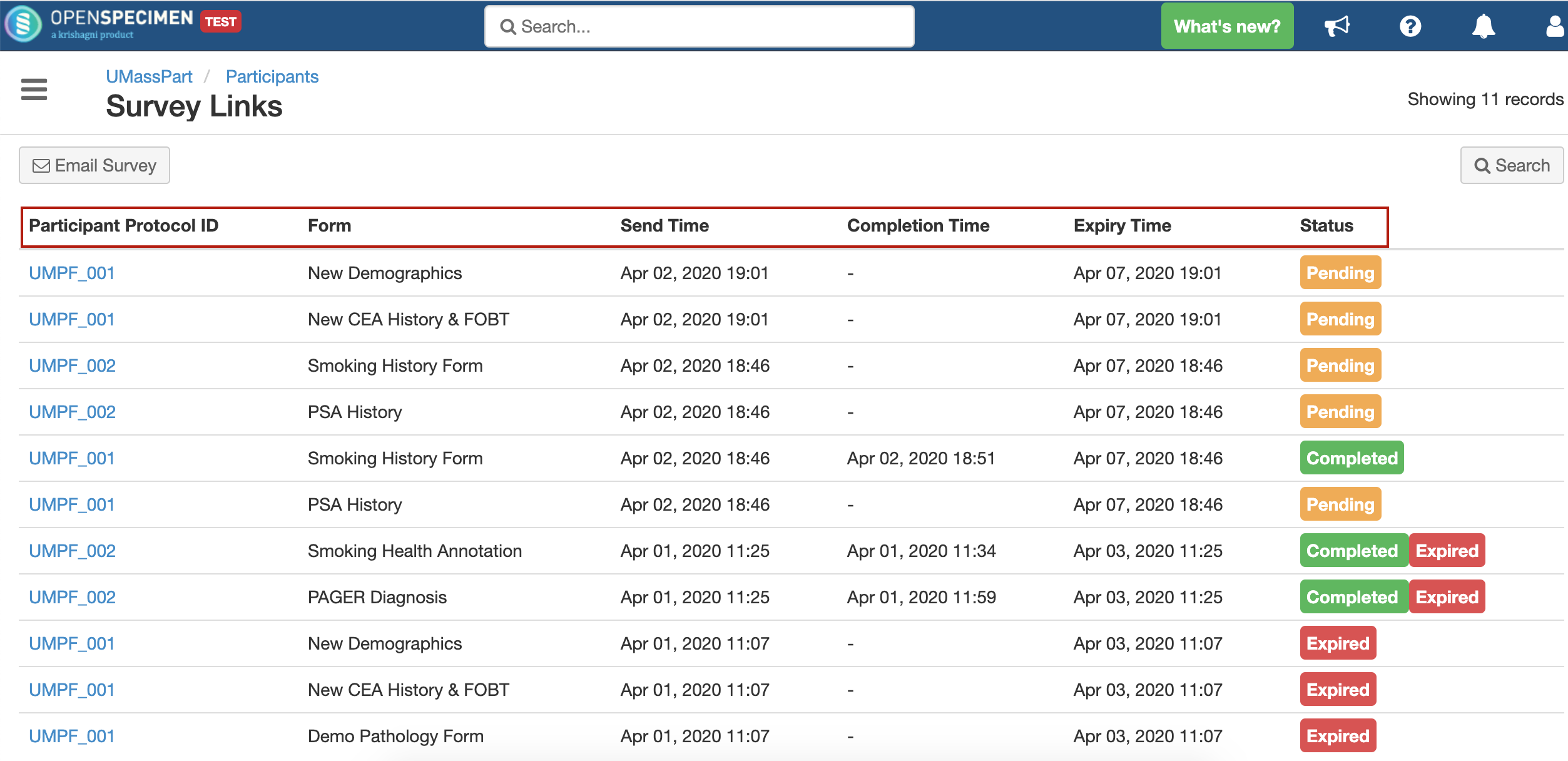
Refer to 'Survey Links' for more details.
The new mobile app runs on any portable android devices like tablets to enable easy data entry of participants and specimens. This also enables users to do offline data entry where they do not have access to network. For e.g when the users are on the field collecting specimens.
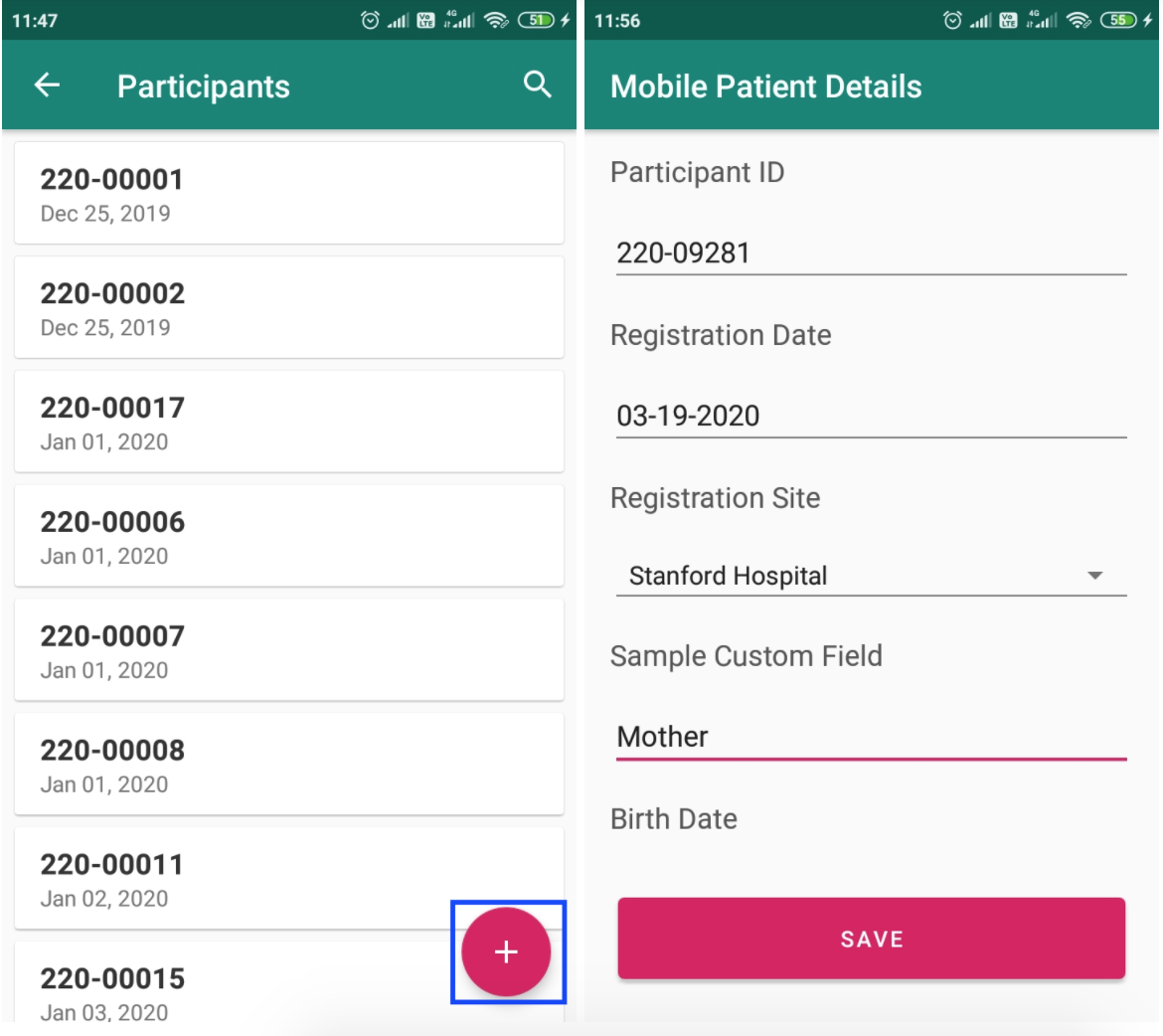
For more details, refer to '/wiki/spaces/DEV/pages/649756703' help pages.
Below are some of the enhancements implemented in v7.0 in REDCap integration plugin:
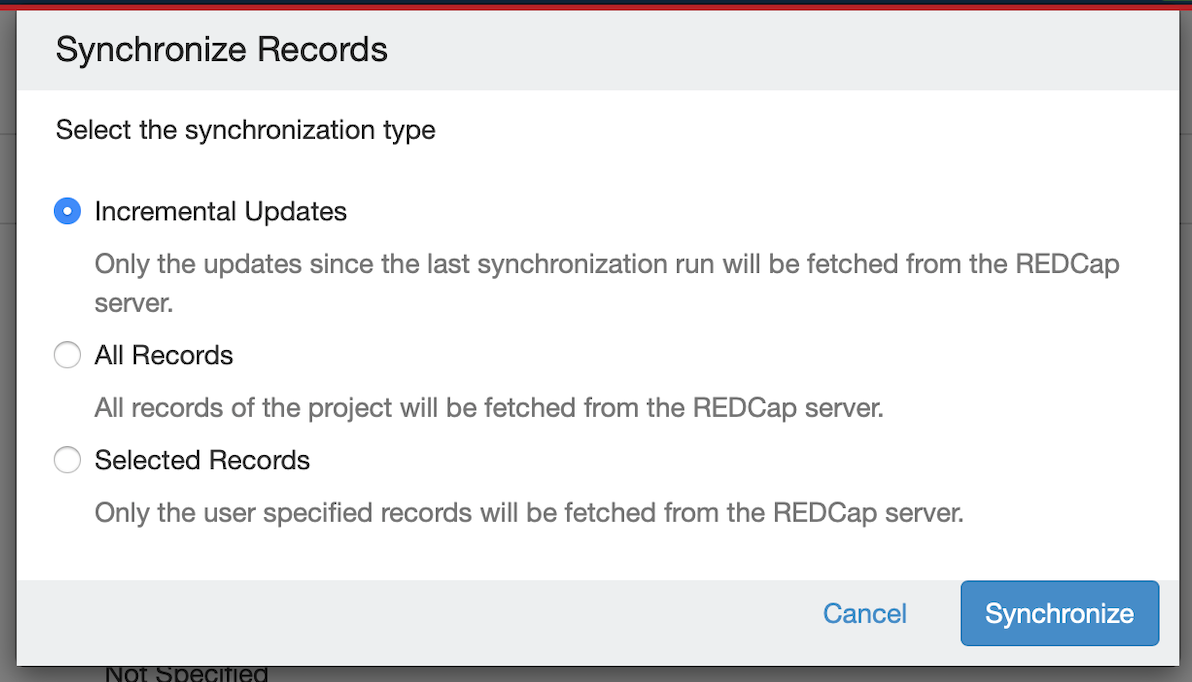
Needs Jira login to view table.
|