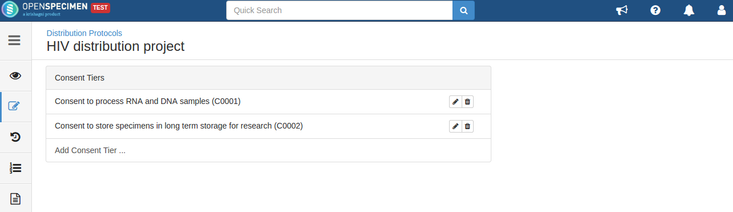
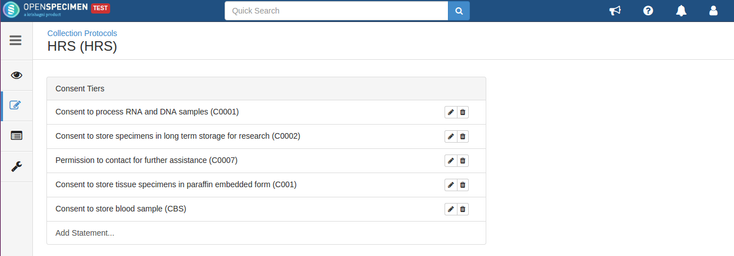
Consent validation
Consents can be validated before distribution by setting required consents at DP and CP levels.
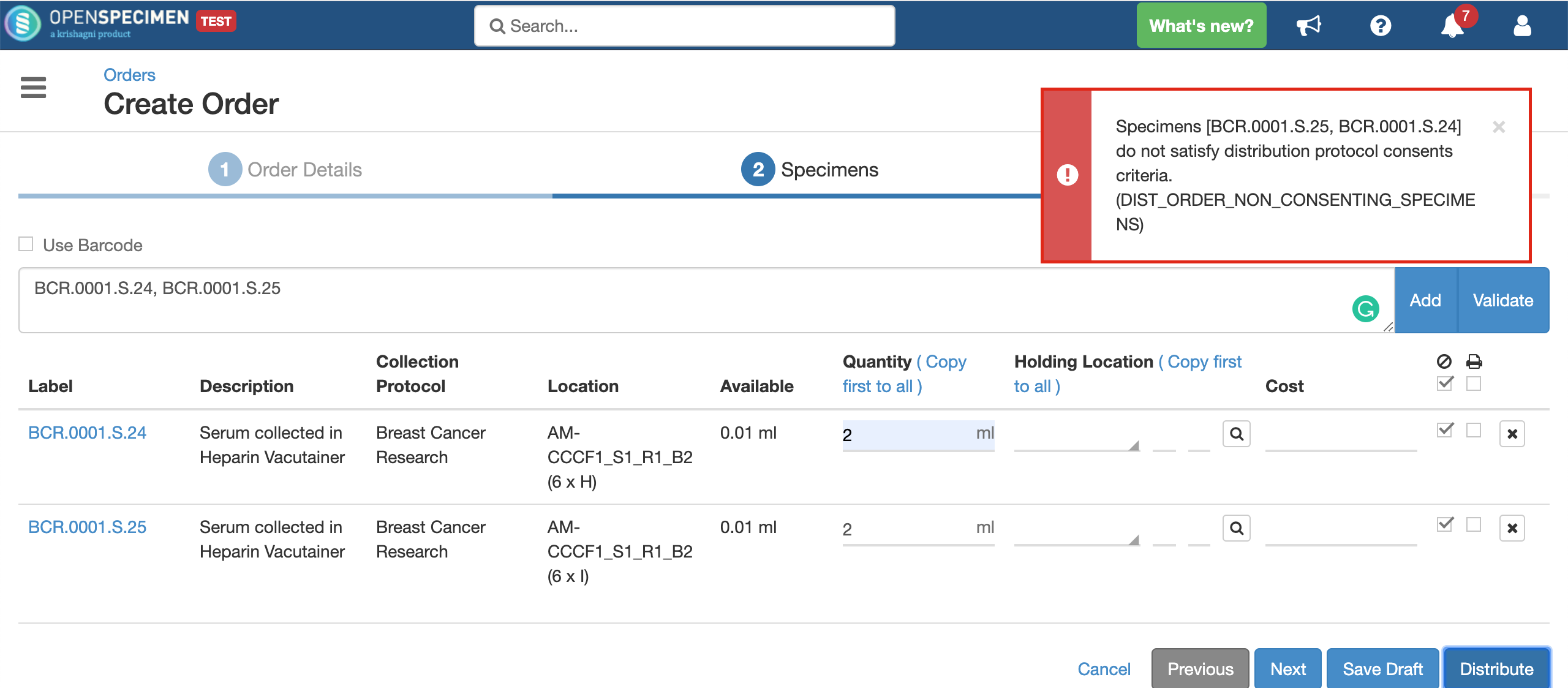
System will allow distributing the specimens only if:
- The consents statements added to DP should match with the CP statements whose specimens are to be distributed (Number of CP consent statements can be greater than DP)
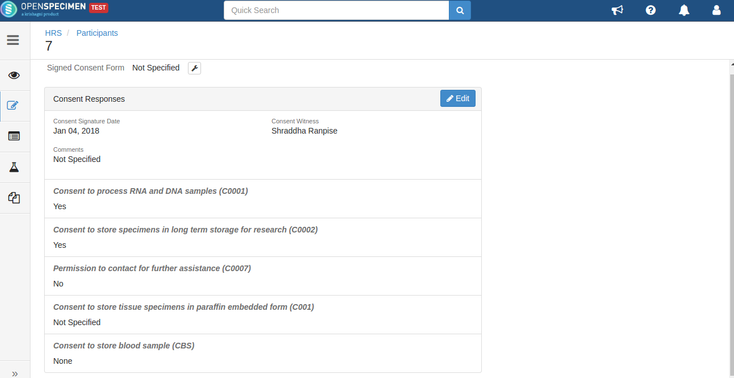
- Consent responses are ‘Yes’ for all these statements under the participants of specimens being distributed.
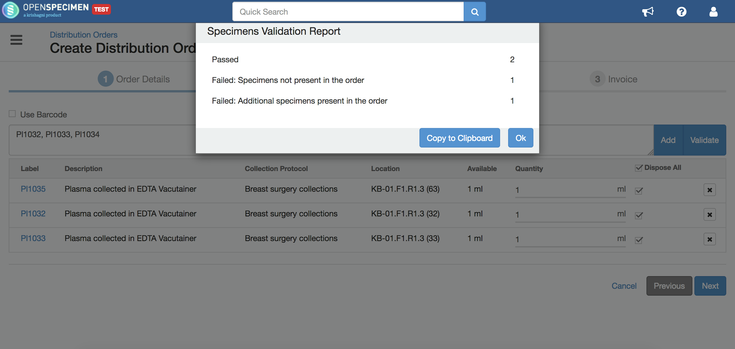
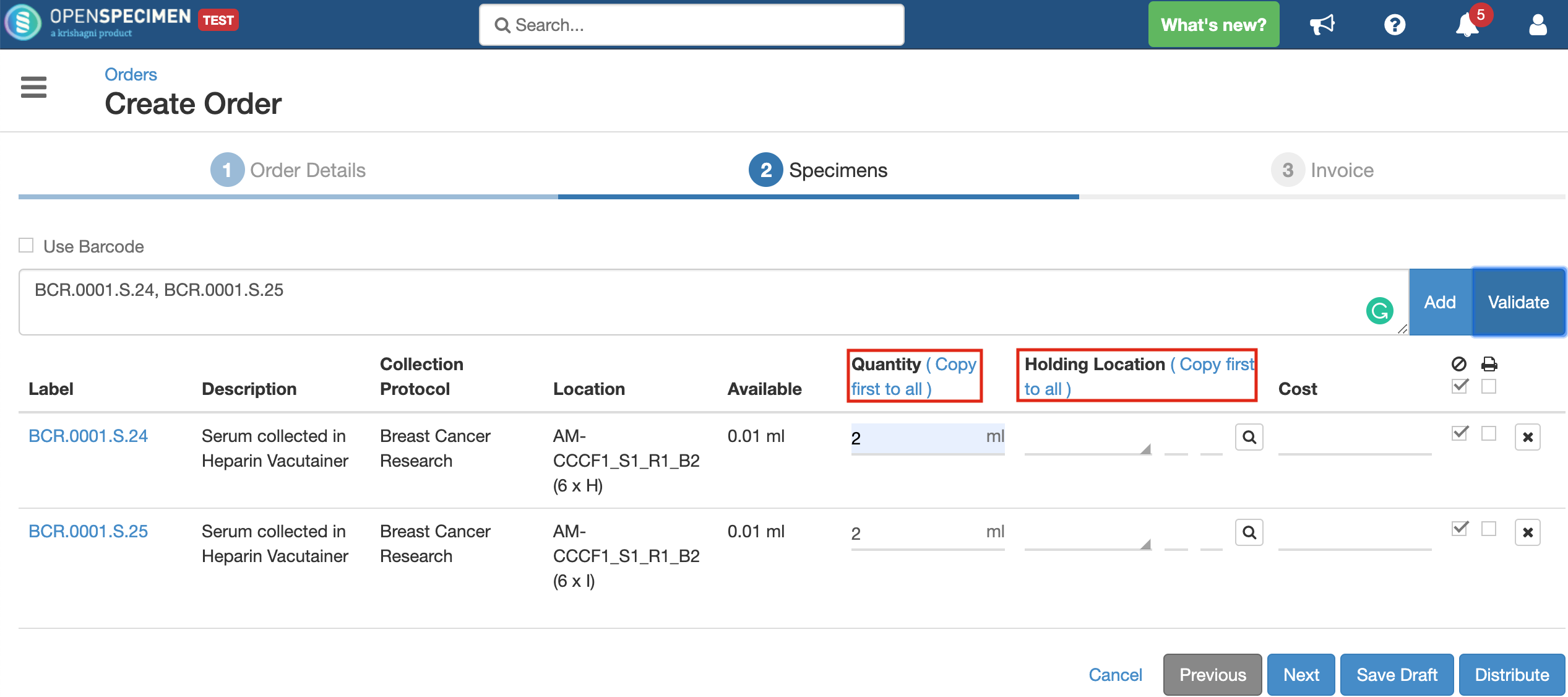
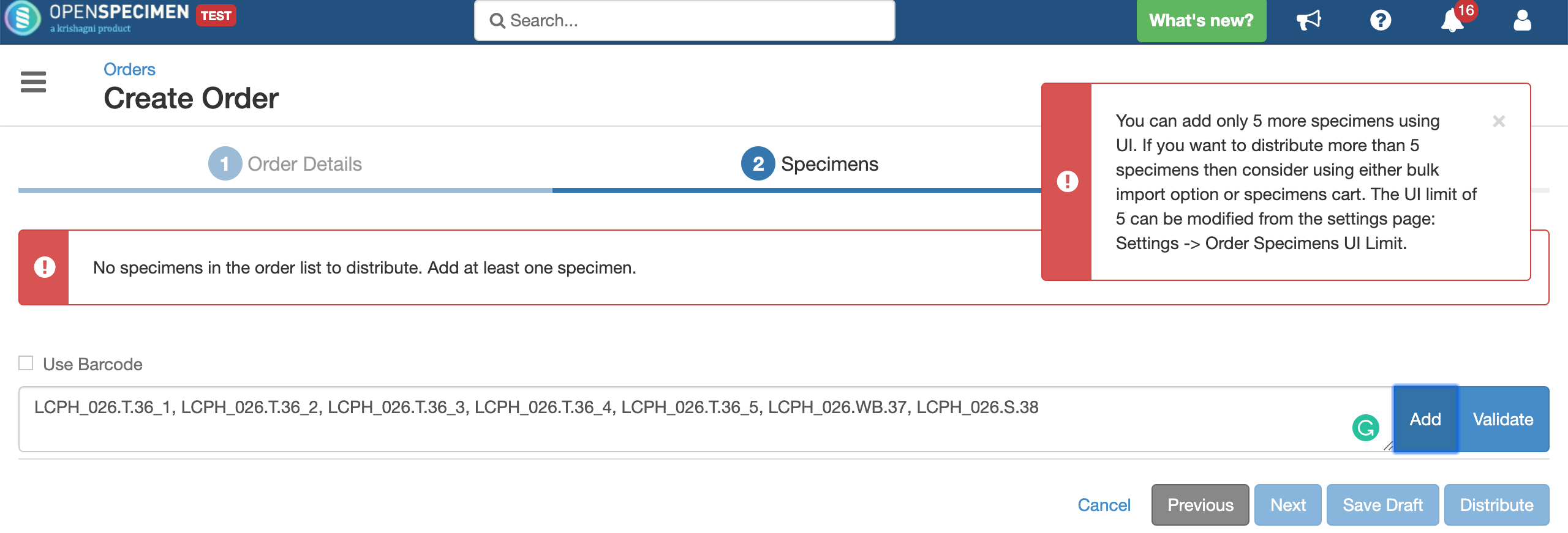
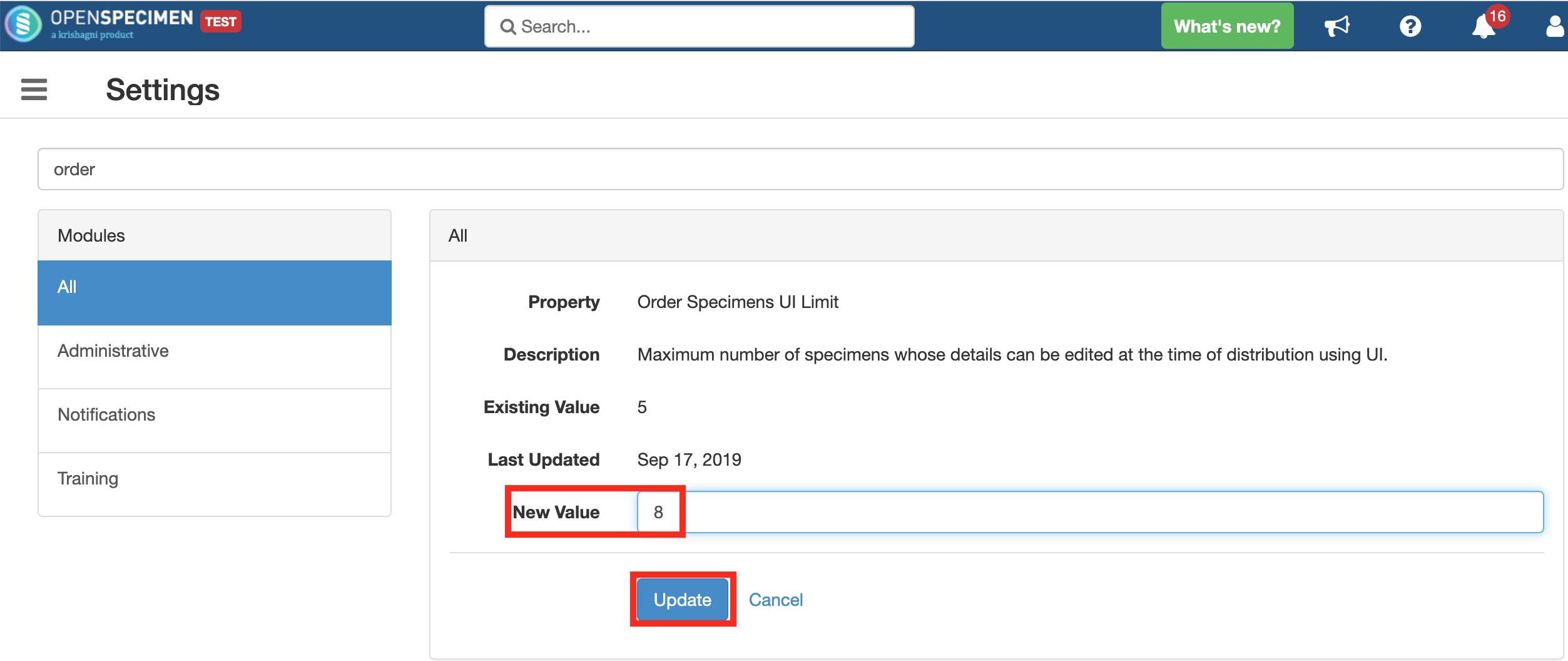
Validating specimens
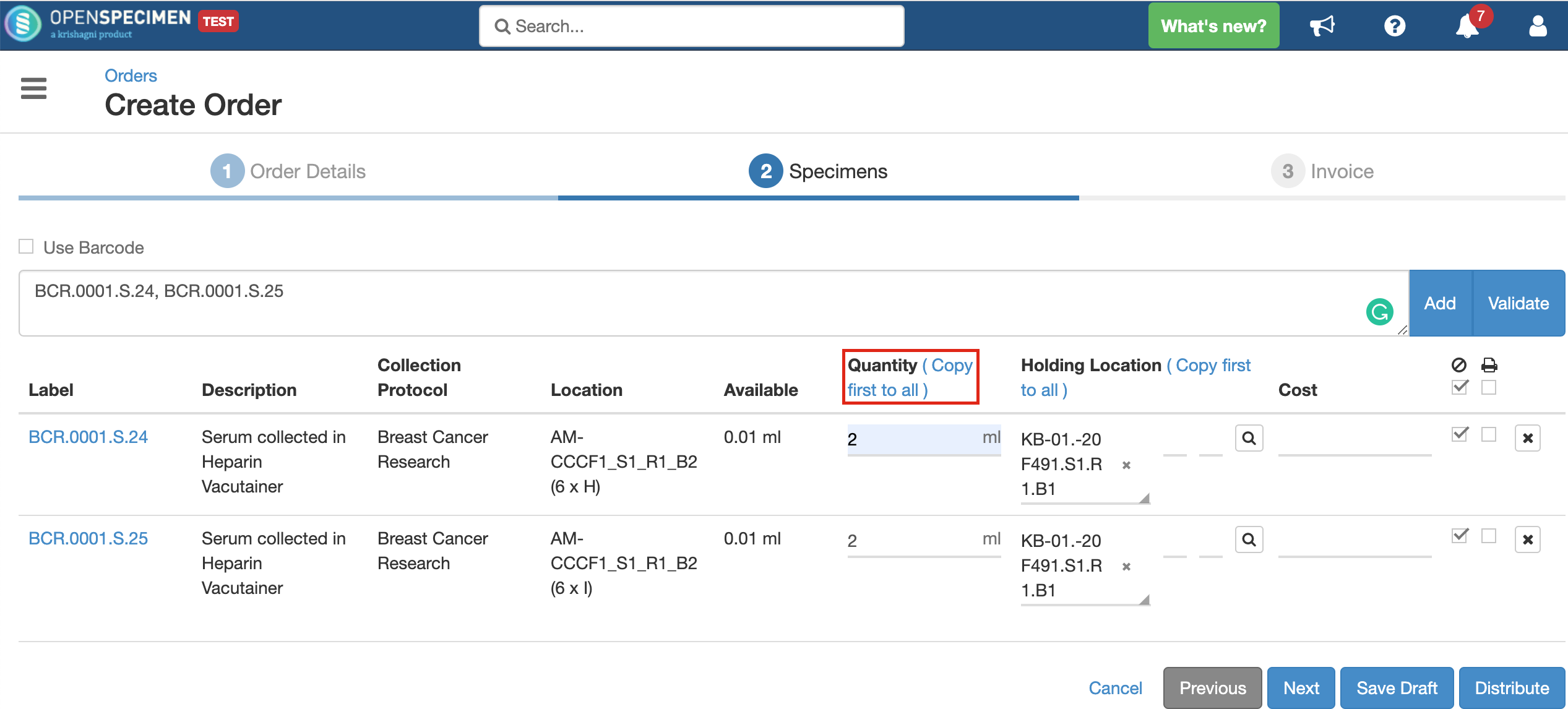
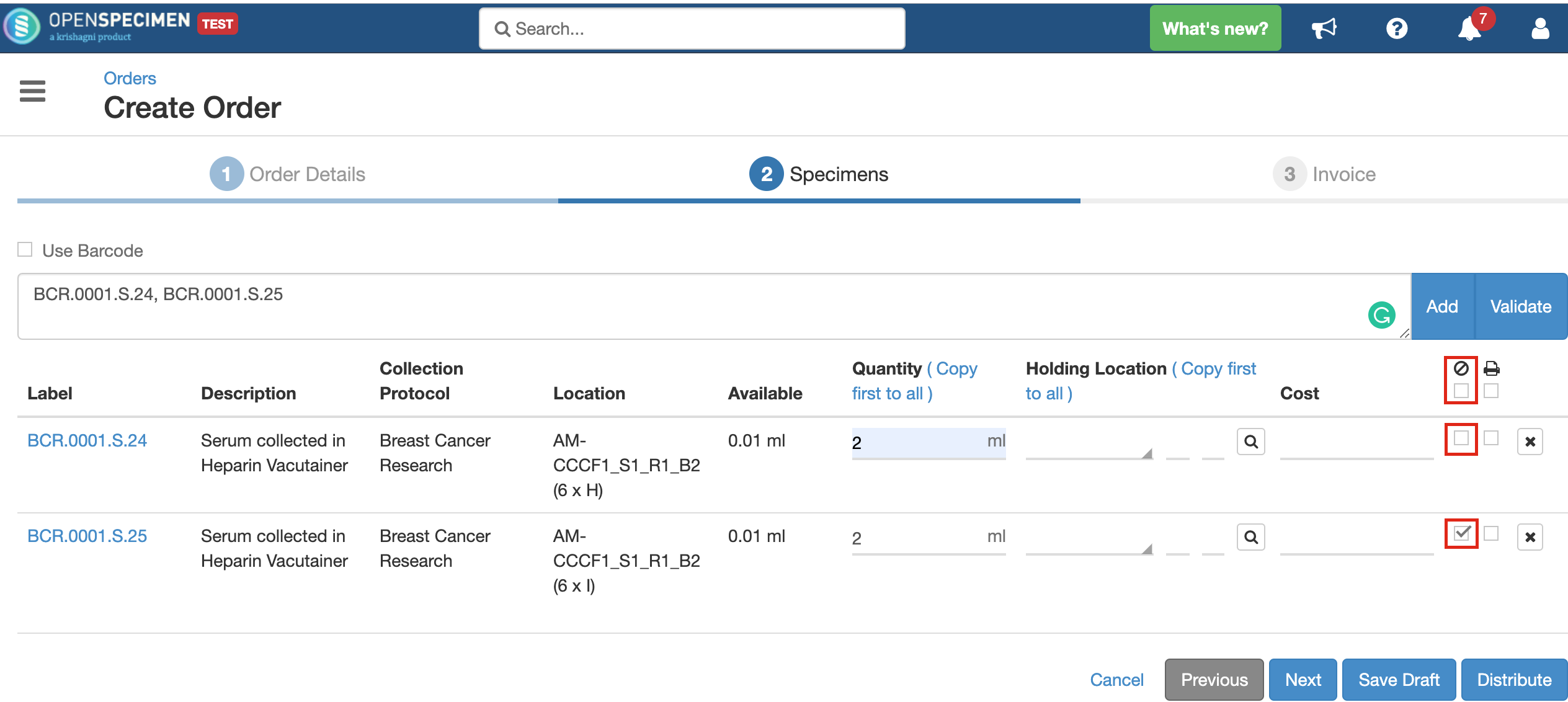
Quantity
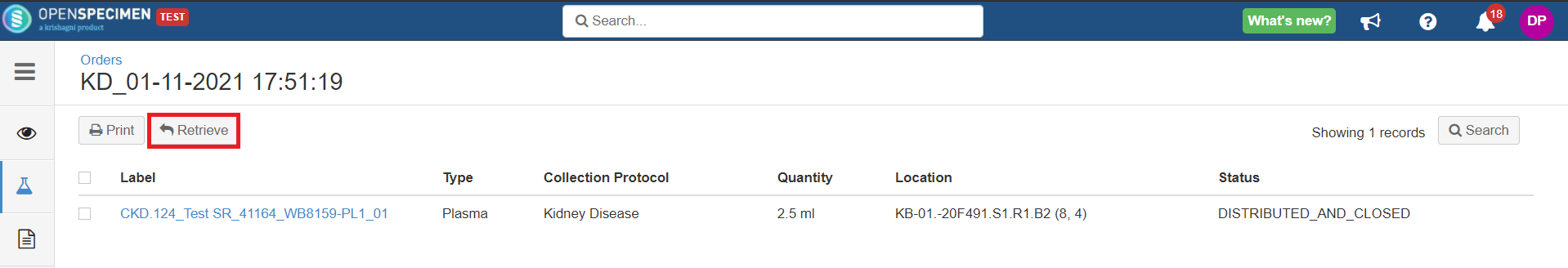
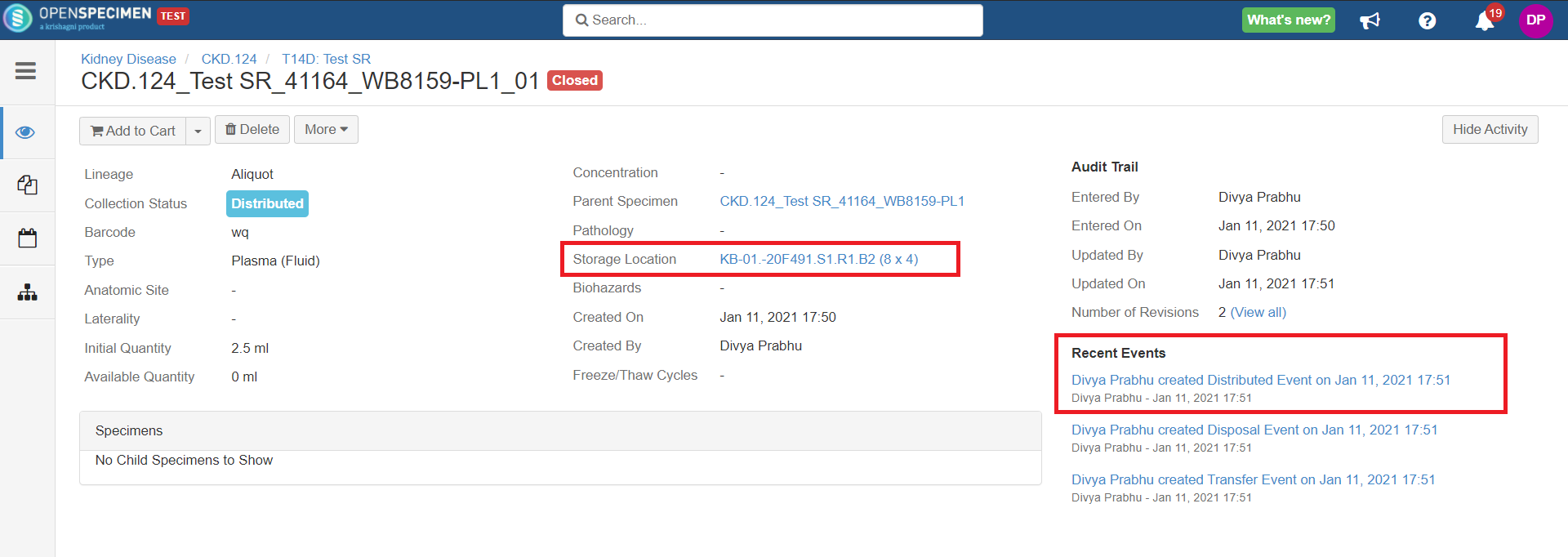
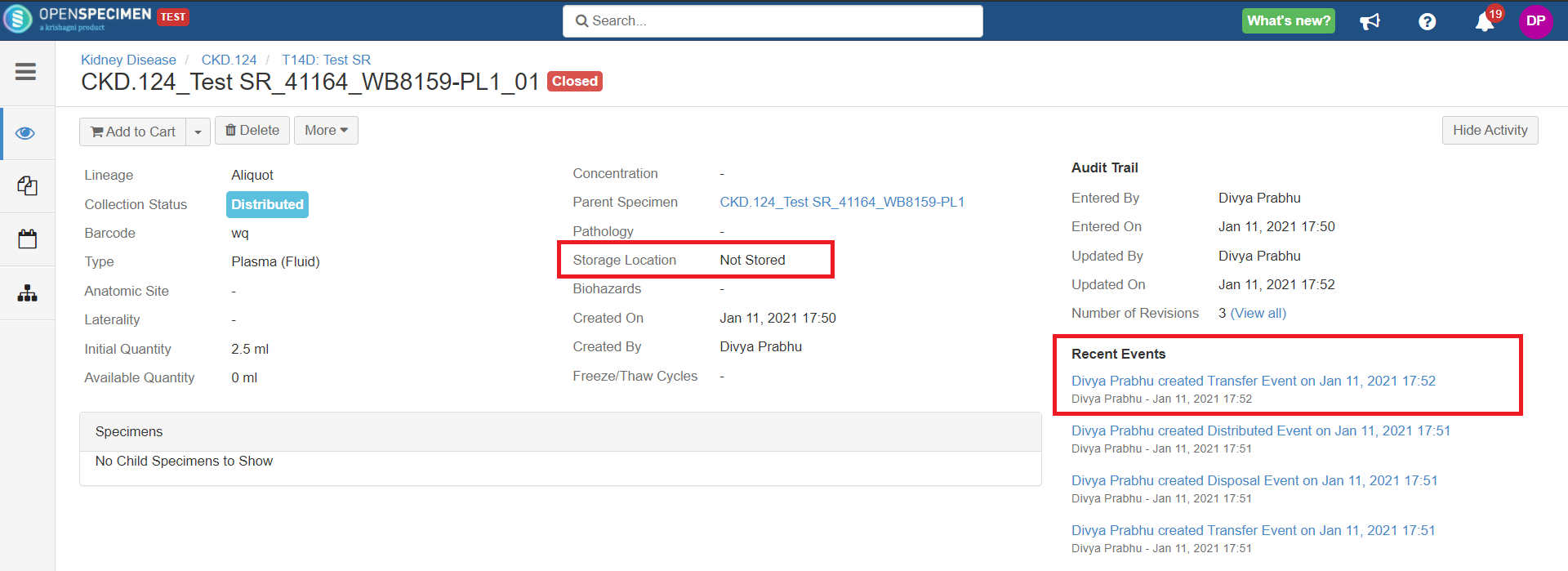
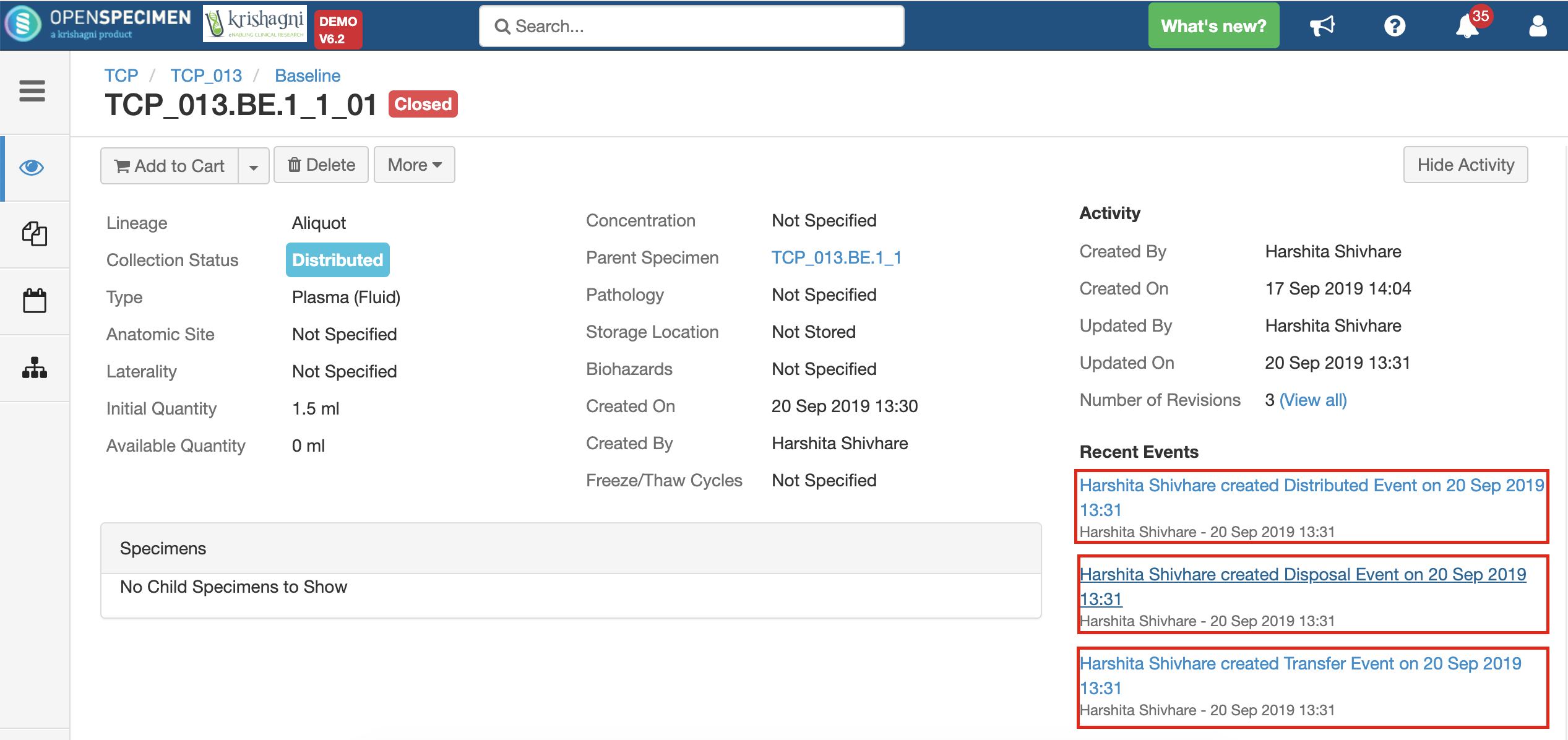
Holding Location
A temporary location where the specimen will be transferred to and stored until the researcher picks it up (retrieves) for his use.
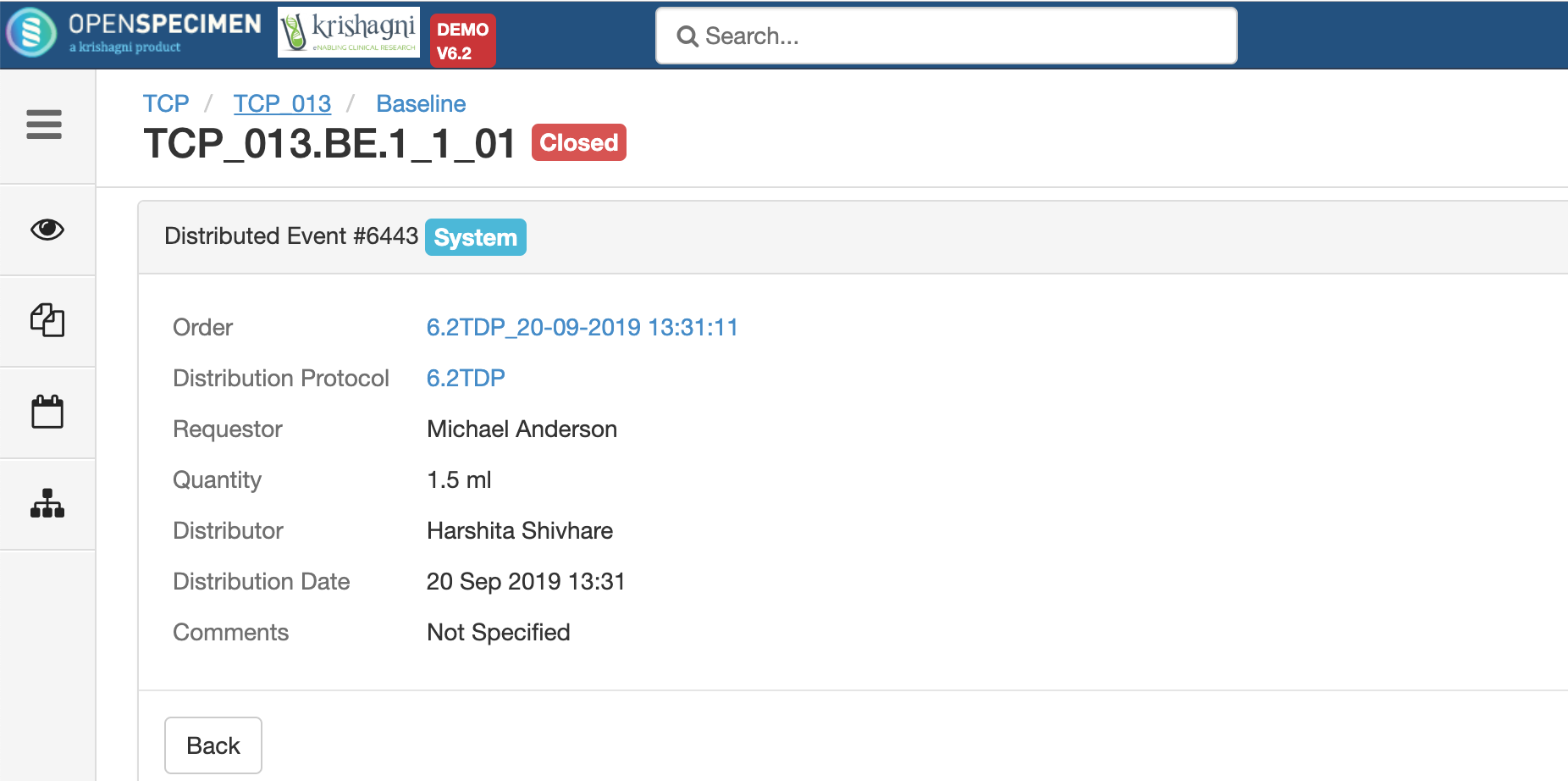
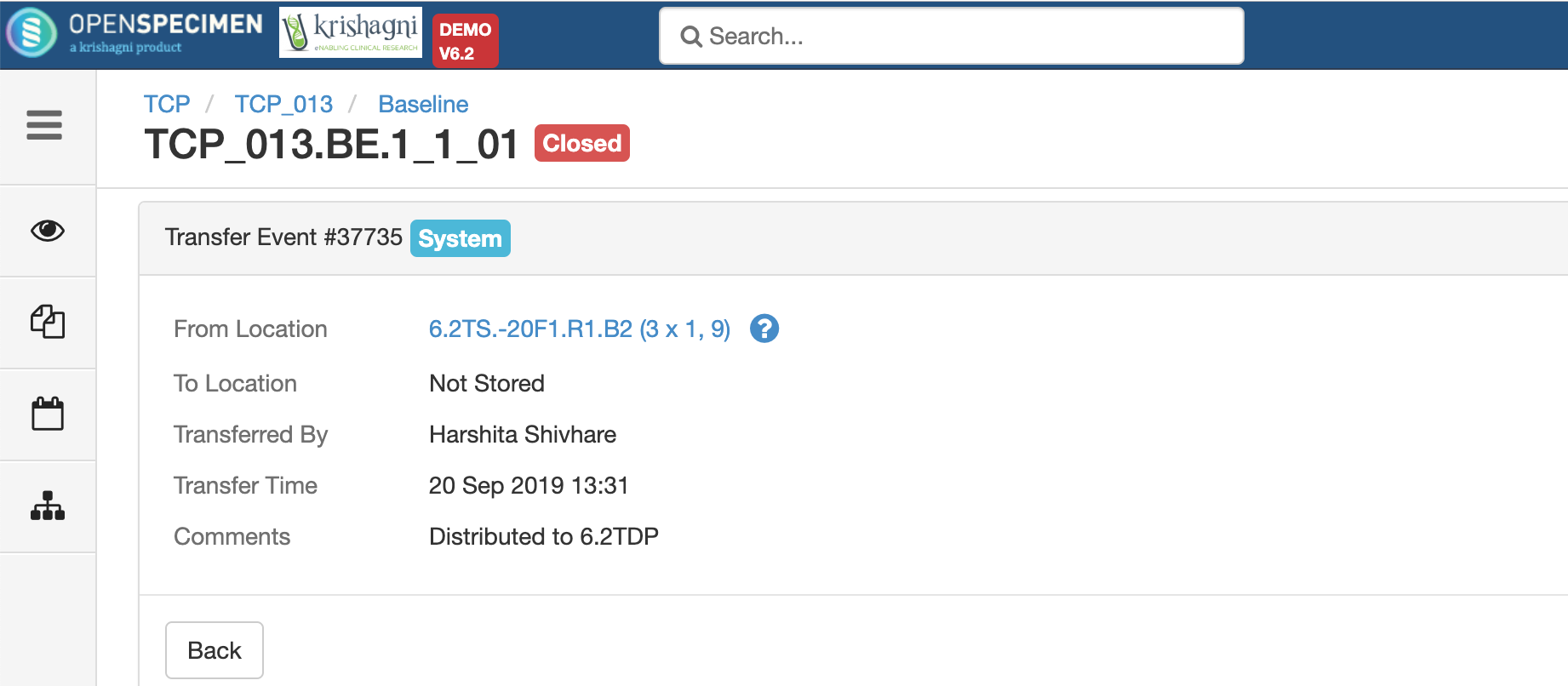
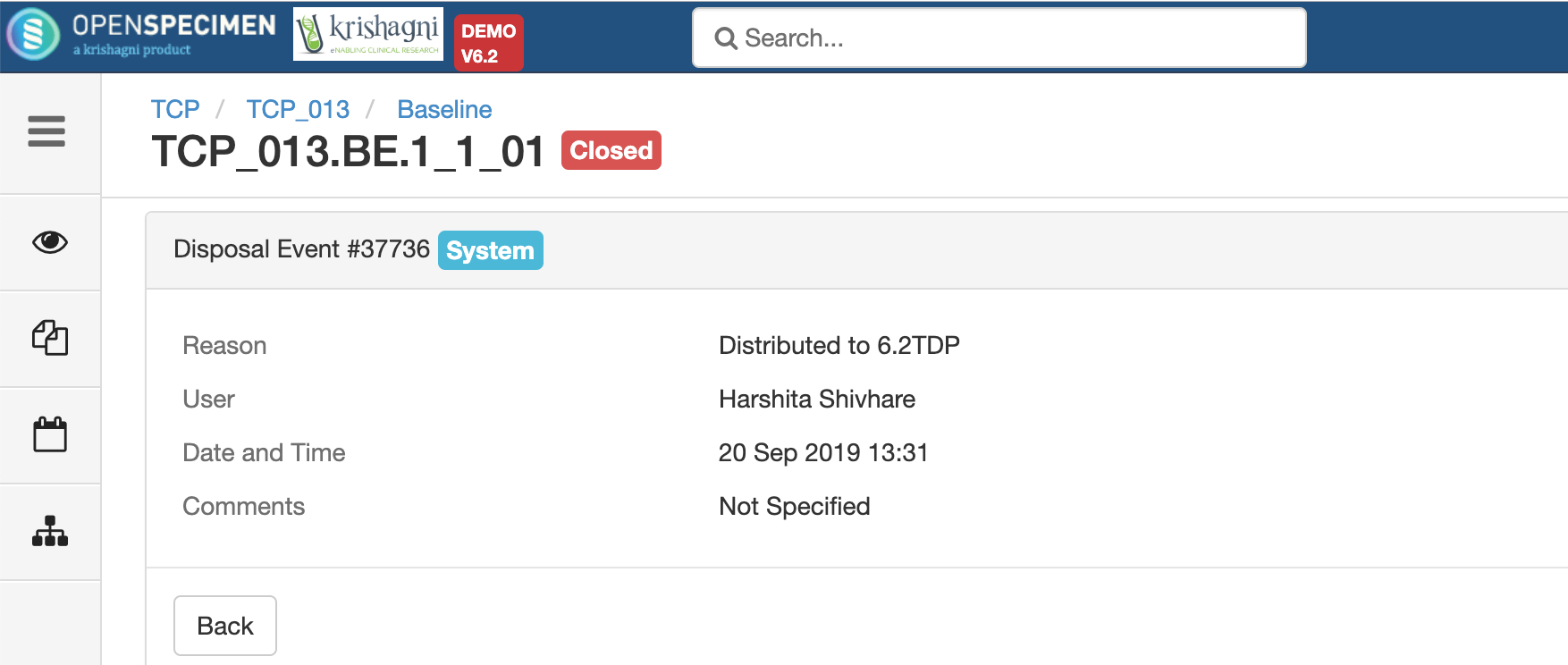
.png?version=1&modificationDate=1600084749191&cacheVersion=1&api=v2&width=735&height=199)